Stem cell therapy drops cost of heart attack aid
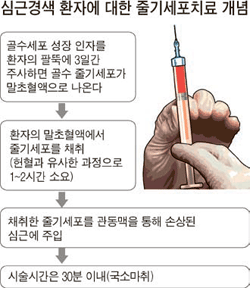
The surgery was performed at the Seoul National University Hospital. At that time, his doctor asked Kim if he was willing to participate in a stem cell clinical test that was being promoted by the hospital, and he agreed.
The surgery involved a procedure of injecting stem cells taken from his blood supply into the damaged heart muscle.
The operation ended after 30 minutes. After six months and two years, the stem cell clinical trial team conducted a magnetic resonance imaging scan of Kim’s heart. It was found to be improved, and it stayed improved during the following checkup period.
A team of professors at Seoul National University Hospital’s cardiovascular center, led by Kim Hyo-soo and Kang Hyun-jae, said yesterday that they requested the Committee for New Health Technology Assessment under the Ministry of Health and Welfare to approve the clinical use of their stem cell therapy in November of last year.
If the ministry accepts their request, which is foreseen to be in the first half of this year, Korea will be the first country in the world to allow doctors to perform surgical operations using stem cell therapy on heart attack victims without legal restrictions.
The introduction of the therapy follows a recent approval by the Korea Food & Drug Administration of the Hearticellgram-AMI, the world’s first stem cell treatment for clinical use for acute myocardial infarction victims, in July of last year. It was developed by FCB-Pharmicell, a Korean biotechnology firm. Industry experts note that there will be competition between the two stem cell therapy and stem cell treatment.
“Stem cell treatment, which is similar to our therapy, is pretty expensive [10 million won, or $8,876, per surgical operation], so we thought that we should commercialize stem cell therapy so that patients are able to get cured [without a burden],” Professor Kim said.
“Once the ministry approves the clinical use of our stem cell therapy development, the price will go down as much as to the 1 million won level.”
Both stem cell treatment and stem cell therapy take stem cells from bone marrow. The Hearticellgram-AMI is injected near the area of the patient’s damaged heart muscle after taking mesenchyme stem cells from his or her marrow.
“Patients express excessive pain when doctors take out stem cells from their marrow,” said Professor Kang.
“Our therapy is different in that we extract stem cells from the blood supply, incubate them, and inject them in the blood vessels.”
By Park Tae-kyun [angie@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)