Invossa scandal continues to build

Invossa is the first drug to use gene therapy to treat osteoarthritis, and was approved for commercialization in Korea in July 2017. The company suspended the sale of Invossa in April 2019 after finding material not previously disclosed and approved by the Ministry of Food and Drug Safety was used in production.
Mitsubishi Tanabe Pharma, a Japanese pharmaceutical company, said that Kolon TissueGene, the subsidiary, knew that an errant cell line was utilized in making Invossa since March 2017. Industry sources believe Mitsubishi Tanabe Pharma came across the information after searching through data it received from Kolon for their partnership.
“When a company signs a licensing agreement with another, hundreds of relevant data are sent to the partnering company,” a Kolon Life Science spokesperson said. Mitsubishi Tanabe Pharma “must have discovered the information while searching through them.”
Mitsubishi Tanabe Pharma is currently involved in a lawsuit with the Kolon affiliate over Invossa licensing. In late 2017, it terminated its agreement with Kolon Life Science for breach of contract. The agreement was worth around 500 billion won ($427.6 million) and signed in 2016.
The Japanese company argues that Kolon failed to notice a location change in the production of sample drugs for U.S. clinical trials.
Shortly thereafter, Mitsubishi Tanabe Pharma filed a complaint demanding the repayment of 26.2 billion won. The agreement gave Mitsubishi Tanabe Pharma exclusive licensing and development rights for Invossa in the Japanese market.
The firm conducted a Short Tandem Repeat (STR) analysis and discovered that a kidney cell line not mentioned in the data submitted to the Korean ministry was used.
STR analysis, developed decades ago, was only used for criminal investigations until early 2000s. It was only after 2010 that the method started being used in developing drugs.
Kolon Life Science has insisted it wasn’t until February that it learned Invossa was made using cell lines not approved by the Ministry of Food and Drug Safety.
The company said its affiliate only mentioned the fact that non-human cells were not found in the results released in March 2017, failing to notice that a kidney cell line was discovered in the analysis.
“The employee must have focused on the fact that there was no problem in producing the drug, not on the fact that an errant cell line was discovered,” a Kolon Life Science spokesperson said Monday.
Lonza conducted the STR analysis to verify that only human cells were used, the company said.
Invossa was granted approval for commercialization in Korea and for clinical trials in the United States on the basis that it used cartilage-originated basis cells.
Industry sources question why Kolon Life Science did not conduct an STR analysis on Invossa even though it had high expectations for the drug. The company believed Invossa could help it gain a competitive edge in the osteoarthritis treatment market and possibly be used to treat other musculoskeletal disorders. Kolon TissueGene’s fate is also heavily dependent on the drug, as Invossa is the only product released by the company.
The company is potentially facing lawsuits from patients around the world over the mislabeling scandal. OhKims Law & Company announced last month that it is seeking people injected with Invossa to pursue a class-action lawsuit. Some industry sources also blame the lack of action on Korean and U.S. authorities.
The Ministry of Food and Drug Safety is visiting Kolon TissueGene for an inspection later this month to find the reason for the decade-long mislabeling, and the FDA suspended the clinical trials for the drug in the United States.
“Based on the data Kolon submitted to us, the ministry could only find that the drug was based on cartilage cells,” a ministry spokesperson said, adding officials are visiting Kolon TissueGene in America on May 20 to follow up.
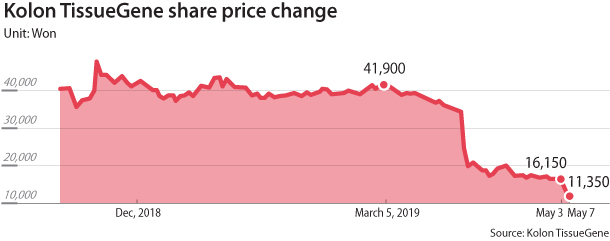
Due to the scandal surrounding Invossa, Kolon TissueGene’s share price on the secondary Kosdaq market fell more than 70 percent from 41,900 won on March 5 to 11,350 won as of Tuesday’s close.
BY LEE SOO-KI, LEE SEUNG-HO, KIM JUNG-MIN AND KO JUN-TAE [ko.juntae@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)