It’s Finland on the line and they need Covid-19 testing
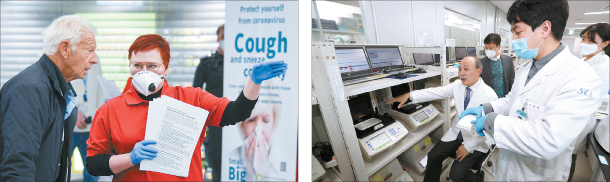
From left: An airport employee at the Helsinki-Vantaa International Airport provides information to a passenger who just arrived. Researchers from Seoul Clinical Laboratories (SCL) are running tests on blood samples. SCL will be in charge of running coronavirus diagnostic tests on patient samples from Finland. [EPA, KIM SANG-SEON]
The package was swiftly loaded onto a minivan and whisked to Seoul Clinical Laboratories (SCL), a bio lab located in Gyeonggi, 40 kilometers (25 miles) south of Seoul.
Helsinki’s Mehilainen, a private health care company, had sent the samples for testing.
When the cargo arrived at the labs at SCL, researchers took them out, extracted the nucleic acid from the specimens, amplified them and conducted an analysis using a technique called RT-PCR.
The results were ready in fewer than four hours.
By 11 p.m, the same day, the results were transmitted back to Finland, just over 12 hours from when wheels touched down at Incheon.
K-bio, which is in the spotlight for its effective and accurate diagnostic kits, is going one step further. Korea is becoming a testing site for possible infections in other countries.
Finland was the first to try.
SCL is located in Heungdeok IT valley, right next to the Gwanggyeo pond, in Suwon, Gyeonggi. At 16,500 square meters (177,604 square feet) in size, it is the biggest single diagnostic lab in Korea. It is furnished with state-of-the-art diagnostic equipment and lab kits.
The diagnostic kits used in the Finland project were products from Seegen and Kogene Biotech, two companies that have signed contracts to supply coronavirus test kits abroad.
Even on a Friday evening when the JoongAng Ilbo visited the lab, it was packed with busy technicians running diagnostic tests.
“We’ve added more equipment and hired new researchers to meet increasing requests coming inside Korea and from abroad,” said Kim Jeong-hwan, the head of human resources at SCL.
Quick and accurate diagnosis
Why would Finland, a highly advanced European Union country, send samples to Korea?
The country reported the first case of the coronavirus on Jan. 29, when a 32-year-old Chinese tourist tested positive for the virus. She was treated in a hospital and sent home on Feb. 5.
While the number of confirmed infections remained relatively flat through early March, confirmed cases started to surge after Finnish nationals back from Italy started exhibiting symptoms.
Through Monday, the number of confirmed patents in Finland climbed to 1,882 with 25 deaths.
Lim Hwan-sub, the head of SCL, thought someone had called the wrong number when the Finnish hospital first contacted his lab on March 20.
“A man named Hartiala who claimed he’s from the Mehilainen hospital called our facility to ask for help in diagnosing their coronavirus samples,” he said.
Finland asked Korea for help as its public health care system struggled to respond to the high demand for tests, as its daily cap was 2,000 samples. While it could import diagnostic kits from Korea, Finland did not have enough RT-PCR equipment to analyze the test results.
Its last option was flying out the samples to a different country.
Korea, China and Japan were considered for testing. It dismissed China due to accuracy concerns. Japan was also marked off since the country was already struggling to keep up with local demand.
Room for more
The first samples from Finland arrived 10 days after the initial call on March 20. SCL was able to send the results through email just seven hours after 100 samples from Finland landed on Korean soil.
The Finish company sent the same number of samples to both Korea and a lab in Estonia. It wanted to see which was better: Korea won.
“After the first round of samples, the Finnish hospital called saying they will cancel the contract with Synlab in Estonia and send all samples to us from now on. They told me our results were more accurate,” said Lim.
Lim is confident that accepting samples from other countries will not cause any problem in diagnosing samples from local Koreans. SCL has increased its diagnostic capacity to cover up to 10,000 samples a day, way above the current daily average of 2,000 to 2,500 samples.
The government also started to respond to similar requests from other countries.
It recently sent a public document to diagnostic labs like SCL, requesting each lab to report to the government when approached with a request from overseas and obtain the government’s permission before accepting offers. This is to prevent any bottlenecks from developing as a result of too many requests.
Seegen Medical Foundation, EONE Laboratories, Green Cross Corporation and SM Lab are some of the local labs with the daily capacity of up to 10,000 samples.
Korean labs have enough capacity to spare for other countries since the number of symptomatic patients and confirmed patients are starting to decline.
It will not be a burden for Korean companies to accept request from other companies, according to the Korean Society for Laboratory Medicine.
BY CHOI JOON-HO [kang.jaeeun@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)