Platform technologies are Holy Grail for vaccine makers
![CHA Vaccine Institute researchers conduct tests at CHA Bio Complex in Pangyo, Gyeonggi. [CHA VACCINE INSTITUTE]](https://koreajoongangdaily.joins.com/data/photo/2021/10/24/0ccbc203-9b7c-4bc1-b12e-6ad26cc22c34.jpg)
CHA Vaccine Institute researchers conduct tests at CHA Bio Complex in Pangyo, Gyeonggi. [CHA VACCINE INSTITUTE]
Viruses change quickly, as the Covid-19 coronavirus has reminded the world. In order to develop new drugs rapidly, “platform technology” is an essential tool for biopharmaceutical companies.
The basic idea of platform technology is that it can be applied to drugs in the development process to maximize their efficacy or convenience for end-users. Local vaccine maker CHA Vaccine Institute's Adjuvant L-pampo and Lipo-pam, technologies that enhance immune system responses, and Hanmi Pharmaceutical's Lapscovery, which helps to prolong the effectiveness of a drug and therefore minimize the frequency of treatment or dosage required, are prime examples.
A platform technology can be applied to various drug candidates, reducing the time and cost needed to develop a new vaccine or treatment.
“Platform technology is an essential tool in order to not only shorten the time to develop vaccines, but also to improve the safety, manufacturing efficacy, ease of storage and transportation, as well as convenience of administration,” Kim Ju-won, a research fellow at the Korea Institute of S&T Evaluation and Planning, said in a report.
“Korea is far behind global companies, but if it keeps trying to develop the technologies and acquires experience, it will be very helpful to the country when it faces another infectious disease in the future.”
The success of Moderna and Pfizer’s mRNA Covid-19 vaccines were the result of platform technology use. It used to take 10 to 15 years to develop a new vaccine. Global bio companies were able to develop Covid-19 vaccines in about a year using their own platform technologies.
The next big thing
CHA Vaccine Institute is developing a therapeutic vaccine for chronic hepatitis B using two platform technologies — Adjuvant L-pampo and Lipo-pam — that contain immune system stimulating materials to increase the efficacy of a vaccine. The drugs made are about 100 times more effective in forming antibodies than those made with conventional technologies, the bio company said.
If CHA succeeds, it will be the first commercialized hepatitis B therapeutic vaccine. The drug is currently undergoing Phase 2 clinical trials in Korea, and the company aims to conclude the trials by 2023.
The company received an S rating for the platform technologies from the Korea Invention Promotion Association. The association rates technologies on an S to D scale, depending on how innovative they are, with S being the highest grade. Only two companies have received S ratings so far, CHA Vaccine Institute and British multinational pharmaceutical company GlaxoSmithKline.
![Vials of the Pfizer Covid-19 vaccine [KIM SUNG-TAE]](https://koreajoongangdaily.joins.com/data/photo/2021/10/24/ac2c3fc1-2522-42b2-a263-84c3b800bd9d.jpg)
Vials of the Pfizer Covid-19 vaccine [KIM SUNG-TAE]
“CHA Vaccine Institute’s two immunity enhancement technologies can be applied to not only drugs targeting infectious diseases, but also many other chronic illnesses and cancer,” said a spokesperson for CHA Vaccine Institute.
The biotechnology company, 38.9 percent owned by CHA Biotech, is also using the technology to develop a herpes zoster vaccine, which It aims to start clinical trials next year.
Hanmi Pharmaceutical has been developing a drug to treat the effects of short bowel syndrome using its Lapscovery technology. On Oct. 12, the company won approval to start Phase 2 clinical trials of the treatment candidate HM15912 in Korea.
Orascovery, another Hanmi platform technology, is helpful in changing injection-type anticancer drugs into orally administered drugs. Many anticancer drugs see reduced efficacy when administered orally; orascovery improves the efficacy, safety and convenience and reduces side effects, the company said.
Daejeon-based LegoChem Biosciences owns antibody-drug conjugate (ADC) platform, which can be applied to biopharmaceutical drugs designed as targeted therapies to treat cancer. Unlike chemotherapy, this kind of treatment spares healthy cells in the treated areas and has been touted as a next-level technology for cancer treatment.
Means of big buck
Profitability is another reason Korean biopharmaceutical companies have been exerting all efforts to develop platform technology. If they succeed in developing one, they can license it for billions of won to big-name pharmaceutical companies.
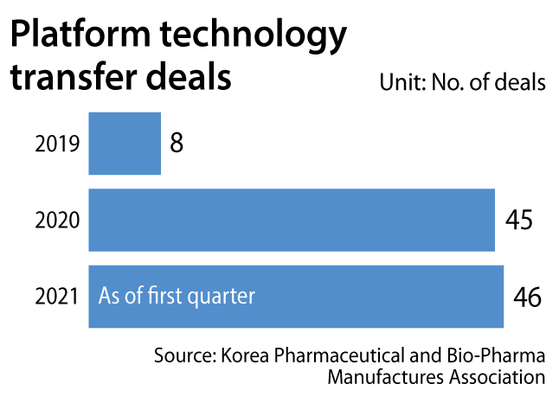
Korean bio companies signed a total of eight deals to transfer their platform technologies in 2019, which increased to 45 in 2020, according to data provided by the Korea Pharmaceutical and Bio-Pharma Manufactures Association. In the first quarter of the year, the number already reached 46.
In March, CHA Vaccine Institute signed a technology transfer deal with Gangnam-based Aston Sci. for the technology transfer of L-pampo. The technology will be used in Aston’s cancer vaccine candidate. The size of the deal is likely to be about 203 billion won including a 2.5 billion won upfront payment, milestone payments and royalities based on the drug’s future sales.
The company also signed a deal with China's CanSino Biologics to license the technology in April 2020. The Chinese vaccine maker aims to use it for its Covid-19 vaccine candidate.
Last year, LegoChem Biosciences signed a license deal worth more than 770 billion won with Britain-based Iksuda Therapeutics for its ADC platform. Since it was founded in 2006, LegoChem Biosciences has signed a total of 10 deals, earning more than 2 trillion won.
Kosdaq-listed Alteogen, which has a technology that can convert intravenous formulations to subcutaneous injection, signed three deals worth about 6.4 trillion won. The technology allows patients to inject treatments themselves instead of being hospitalized for intravenous treatment.
“Demand for Alteogen’s platform is likely to increase further,” said Lee Dong-gun, a senior researcher at Shinhan Investment. “It is anticipated that the company would win big technology transfer deals in the near future.”
BY SARAH CHEA [chea.sarah@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)