[NEWS ANALYSIS] Second Battle of the Botoxes breaks out in Korea
![[SHUTTERSTOCK]](https://koreajoongangdaily.joins.com/data/photo/2022/04/13/e0803ac5-330b-44d3-a681-0a21fcbedd70.jpg)
[SHUTTERSTOCK]
A similar complaint pitted Medytox against Daewoong Pharmaceutical in 2019, which was eventually won by Medytox. The current legal battle is likely to clip the wings of Hugel, Korea’s largest botulinum toxin maker in terms of market share. Hugel was planning to enter the U.S. market this year.
On March 30, Medytox filed a complaint with the U.S. International Trade Commission (ITC) against Hugel, Hugel America and Croma-Pharma GmbH, saying they violated section 377 of the U.S. Tariff Act by stealing botulinum toxin strains and production methods to develop its botulinum toxin product.
Based in Vienna, Croma-Pharma is Hugel’s European partner. Hugel America is a U.S. joint venture established between Hugel and Croma-Pharma.
“Hugel is trying to export its botulinum toxin product developed with strains and a manufacturing process that it stole from Medytox,” Medytox said in a statement. “The ITC should initiate an investigation into this, and must ban imports of the product [to the U.S.]. We also strongly urged the U.S. government body to prohibit the sale of the product already exported to the U.S. market, as well as any commercial and marketing activities.
“This is a justifiable and right act for us to protect the intellectual property that we achieved through lengthy research and investment.”
Medytox hired the Los Angeles-based law firm of Quinn Emanuel Urquhart & Sullivan to represent it and said an unnamed investment firm will pay all legal expenses. If the ITC rules in favor of Medytox, the company will divide any settlement with the investment film.
![Hugel's Botulax botulinum toxin products [HUGEL]](https://koreajoongangdaily.joins.com/data/photo/2022/04/13/57835ff0-90d4-48ab-a169-b17cb298f877.jpg)
Hugel's Botulax botulinum toxin products [HUGEL]
Botulinum toxin, popularly known by the brand name Botox — developed by Allergan, which was acquired by AbbVie in 2019 — is a substance that paralyzes muscles and is frequently used by surgeons to reduce wrinkles.
The current battle comes three years after Medytox filed an almost identical complaint against Daewoong Pharmaceutical.
In January 2019, Medytox and AbbVie, its U.S. partner, complained to the ITC that Daewoong Pharmaceutical’s botulinum toxin Nabota was made with stolen trade secrets. After two years, the ITC ruled in favor of Medytox and ordered Daewoong to halt exports to the United States for 21 months.
The 21-month import ban was nullified in February 2021 after Medytox and AbbVie reached an agreement with California-based Evolus, the global distributor of Daewoong’s Nabota. The deal gave Evolus a license to produce and distribute botulinum toxin products using Medytox technology, and in exchange, Evolus offered a license fee of $35 million to AbbVie and Medytox, to be paid over a two-year period. Evolus also agreed to pay royalties to AbbVie and Medytox, proportional to Nabota’s U.S. sales, for 21 months. Medytox also receives royalties from Evolus for Nabota sales outside the U.S. market, including in Europe, Canada, Russia and Japan.
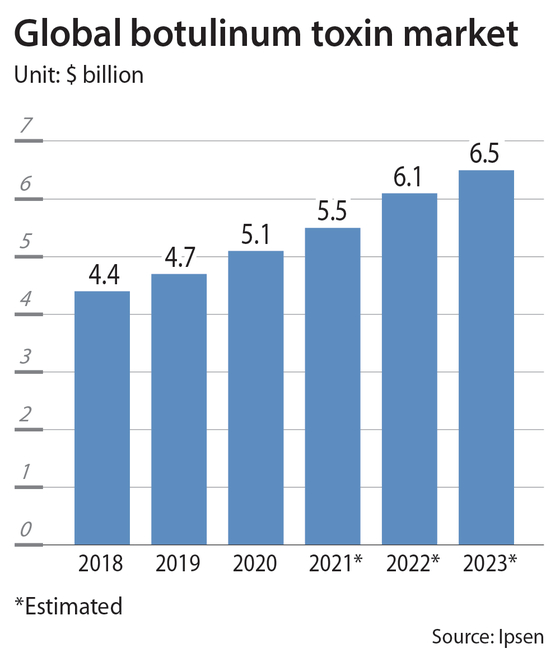
War over bacteria
Botulinum toxin is one of the most poisonous biological substances known to man, a neurotoxin produced by the bacterium Clostridium botulinum. The main issue in these legal fights is where the companies got the bacterial strains.
Medytox was the first Korean company to succeed in mass-producing a botulinum toxin in 2006. It started selling the product in 2014 under the name Meditoxin. Hugel joined the market in 2010 with its product Botulax and Daewoong in 2013 with Nabota. Nabota obtained U.S. Food and Drug Administration (FDA) approval in 2018, becoming the first Korean botulinum toxin product to enter the U.S. market.
According to Medytox, the strains all trace back to a single individual: Yang Gyu-hwan, who served as minister of food and drug safety in the Kim Dae-jung administration. Medytox claims that Yang first developed a strain at a lab at Wisconsin University when he was studying there. Yang brought that to Korea for further research in 1979. He passed it on to Jung Hyun-ho, the current CEO of Medytox, who was a student of Yang's at KAIST.
"Yang brought the strain into the country in his bag," a spokesperson for Medytox told the Korea JoongAng Daily. "Although that kind of activity is illegal now, it was not legally prohibited at the time."
Medytox says local botulinum toxin makers stole the strains from it, although how Daewoong and Hugel got the strain is not clearly known.
Daewoong said it found its strain of botulinum toxin in Yongin, Gyeonggi, while Hugel said it got its from a can of beans that passed its expiration date found in Gapyeong, Gyeonggi.
In December 2020, the ITC affirmed Daewoong’s misappropriation of Medytox’s manufacturing process, but it did not acknowledge Medytox's strain as a trade secret. The ITC announced a 10-year import ban in its initial determination but reduced it to 21-months for that reason.
![Medytox's Meditoxin botulinum toxin products [MEDYTOX]](https://koreajoongangdaily.joins.com/data/photo/2022/04/13/4e63b166-9b4b-4ab9-899f-2f0499d7eac8.jpg)
Medytox's Meditoxin botulinum toxin products [MEDYTOX]
Hugel’s headache
Medytox’s suit has become a big obstacle to Hugel, the company that was just about to start selling in the U.S. market.
Hugel is waiting for marketing approval from the FDA for its botulinum toxin products. The FDA concluded site inspections of the company’s manufacturing plant in Chuncheon, Gangwon, in August last year. Hugel has been expecting final FDA approval within the first half of the year.
The United States is the world’s biggest botulinum toxin market with around 60 percent market share. Its market size is estimated at about $3 billion, according to a report from Kyobo Securities.
On April 1, Hugel issued a statement saying Medytox is trying to “hold us back,” and it will take “all legal steps to fight against it.”
“There is no evidence or circumstances that back up Medytox’s nonsense argument in any process of developing our botulinum toxin product,” Hugel said. “The action by Medytox, which attempts to harm another company’s reputation based on false information and hinder its growth, seriously impinges on the development of the industry and benefits to the country.”
Hugel shares plunged 13.23 percent on April 1, the day Medytox announced that it filed its complaint, and fell another 9 percent the next day. They closed at 120,000 won ($98) Wednesday, down 14 percent compared to March 31.
Experts say Hugel is in a weak position since Medytox has experience with an ITC complaint on exactly the same issue.
Expenses are another issue. Hugel is expected to spend a lot on the legal fight, while Medytox has another company paying its legal bills.
“In the case of Daewoong Pharmaceutical, it said it spent around 35 billion won in 2020 alone for the fight [with Medytox],” said Kim Jeong-hyeon, an analyst at Kyobo Securities. “It is very hard to predict the expenses, but it is expected that Hugel will have to spend around 22 billion won in 2022 and 10 billion won in 2023.”
Established in 2001, Hugel sells its botulinum toxin products in a total of 28 countries. It recently obtained use approvals from seven European countries. The company has been Korea's No. 1 botulinum toxin manufacturer in terms of sales since 2016, followed by Medytox and Daewoong Pharmaceutical.
BY SARAH CHEA [chea.sarah@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)