Got a new vaccine? Korea can make it for you.
![An SK Bioscience researcher conducts tests to develop a Covid-19 vaccine at the company's lab. [SK BIOSCIENCE]](https://koreajoongangdaily.joins.com/data/photo/2021/07/04/fa717710-1cd4-46d3-88de-560fed4f5ae4.jpg)
An SK Bioscience researcher conducts tests to develop a Covid-19 vaccine at the company's lab. [SK BIOSCIENCE]
Making Korea a global hub of vaccine production, one of the goals laid out by President Moon Jae-in, is very slowly becoming a reality with domestic biotech companies’ growing presence in contract manufacturing organization (CMO) market.
Although Korea has yet to make a Covid-19 vaccine of its own, the pandemic delivered local biotech companies an opportunity to prove their potential. Korea’s goal of becoming a global hub of vaccine production has two specific tracks: Stabilizing vaccine supply through mass production and developing its own Covid-19 vaccine.
A CMO is a company that provides drug manufacturing services in the pharmaceutical industry on a contract basis.
On June 28, Huons Global announced that its consortium has won a contract manufacturing deal to produce Russia’s Covid-19 vaccine Sputnik Light. It will become the fifth coronavirus vaccine to be made in Korea.
In late July, 2020, SK Bioscience was selected as one of the global suppliers for Cambridge, Britain-based AstraZeneca’s Covid-19 vaccine. It was the first time a Korean bio company had been selected as the supplier for a Covid-19 vaccine.
The next month, the vaccine subsidiary of SK Group was also named as a contract manufacturer for American bio firm Novavax’s NVX-CoV2373 coronavirus vaccine.
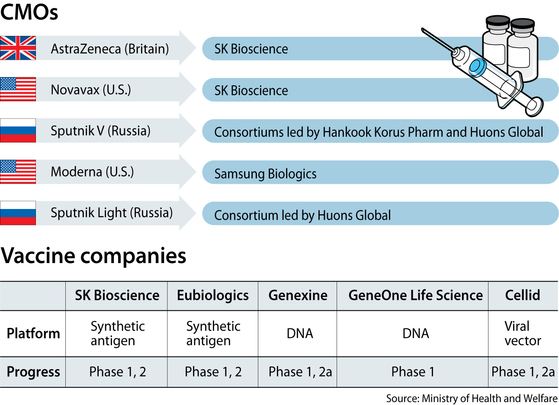
Pharmaceutical companies Hankook Korus Pharm and Huons Global are currently the domestic CMOs for Russia's Sputnik V. Sputnik V was the world's first Covid-19 vaccine to gain regulatory approval, which came out in August last year and is used in more than 60 countries around the world, mostly in South America, Africa and the Middle East. Sputnik V hasn't requested approval for use in Korea yet.
In late May, Samsung Biologics, the biotech arm of Samsung Group, signed a deal with Moderna to manufacture the U.S. pharmaceutical company’s Covid-19 vaccines in Korea. The company will manufacture Covid-19 vaccines at its production plant using Moderna's raw materials produced outside Korea.
“More domestic companies are manufacturing Covid-19 vaccines, and this proves that Korea’s position in the CMO market has reached a global level,” said analyst Na Seung-doo from SK Securities. “In particular, more countries have been approving Sputnik V vaccines around the world, but production delays are causing a shortage.”
“It could be a great and new opportunity for Korea.”
The global CMO market, which was valued at about $118.8 billion in 2020, is estimated to reach $175 billion by 2025, according to a report by SK Securities.
Samsung Biologics is currently the largest company in the global CMO market in terms of capacity. The company has an annual CMO capacity of 364,000 liters (96,200 gallons) per year, which accounts for 28 percent of the total in the global CMO market. Switzerland-based Lonza is the second largest, with 260,000 liters of capacity, while Germany's Boehringer Ingelheim is third with 240,000 liters per year.
The company’s market share in the CMO market is likely to grow further as soon as construction of its fourth plant is completed, which is scheduled to be in August 2023.
The factory, which will have an annual capacity of 256,000 liters, which Samsung claims is the highest figure for a single bio production plant in the world, will raise the company’s annual production capacity to 620,000 liters per year. It is equivalent to nearly 30 percent of the global contract manufacturing market for bio pharmaceuticals.
However, some experts argue that it’s too early to call Korea a hub for vaccine production. Korea lacks crucial vaccine technology like mRNA, or messenger ribonucleic acid, and hasn't succeeded in developing Covid-19 vaccines.
mRNA technology induces an immune response by injecting antigen genes into the body to create spike proteins. The most successful Covid-19 vaccines so far, such as Moderna and Pfizer, are mRNA vaccines.
“It's not time to praise ourselves by only seeing what we can do," said Lee Seung-kyu, vice chair of KoreaBIO. "Since there is no mRNA vaccine technology in Korea yet, it is important to acquire it quickly through the technology transfer of various vaccines."
“mRNA technology is a great clue that can develop not only Covid-19 vaccines, but also treatment for infectious diseases," Lee added. "At this point, it is highly probable that mRNA will be the platform for next-generation treatment."
![Samsung Biologics' third plant in Songdo, Incheon [SAMSUNG BIOLOGICS]](https://koreajoongangdaily.joins.com/data/photo/2021/07/04/b8331343-651c-4df6-89da-48706881a19c.jpg)
Samsung Biologics' third plant in Songdo, Incheon [SAMSUNG BIOLOGICS]
Samsung Biologics on June 1 revealed a plan to add more facilities that could produce “mRNA vaccine drug substances” at its Songdo, Incheon, factories. The goal is to start mass production from the first half of 2022.
In late May, GeneOne Life Science and Hanmi Science announced a partnership to cooperate in building mass production facilities for mRNA vaccines. GeneOne is already developing a Covid-19 DNA vaccine of its own and was one of the first local companies to receive a government nod to undertake clinical trials for a coronavirus vaccine.
A few local companies have been working to develop Covid-19 vaccines. Currently, five Korean companies have received approval to undertake clinical trials for coronavirus vaccines: SK Bioscience, Cellid, GeneOne Life Science, Genexine and Eubiologics, according to the Ministry of Health and Welfare.
On June 28, SK Bioscience filed an investigational new drug (IND) application for phase 3 clinical trial of its own vaccine candidate, GBP510, to the Ministry of Food and Drug Safety.
It is the first phase 3 application for a Covid-19 vaccine developed in Korea.
The bio company also has another vaccine candidate, NBP2001, and said phase 3 clinical trials will proceed on one of the two candidates after it has finished testing which is the most probable one.
It aims to commercialize the vaccine in 2022, according to a spokesperson for SK Bioscience.
Cellid said it aims to run phase 3 clinical trials for its vaccine candidate by September this year.
The Korean government on June 25 promised to strengthen support for homegrown Covid-19 vaccine development so that the possible candidates can enter the final stage of clinical trials by the end of the year.
It will offer companies some services to help expedite the process, such as one-on-one customized counseling. The Ministry of Food and Drug Safety will start operating a central Institutional Review Board to reduce the work involved in receiving IRB approval from different medical institutions.
![President Moon Jae-in, center, and SK Group Chairman Chey Tae-won, left, look at vaccine development facilities during a visit to SK Bioscience's factory in Andong, North Gyeongsang. [JOINT PRESS CORPS]](https://koreajoongangdaily.joins.com/data/photo/2021/07/04/79933ab7-c570-496a-b536-6ae0e6dcac1a.jpg)
President Moon Jae-in, center, and SK Group Chairman Chey Tae-won, left, look at vaccine development facilities during a visit to SK Bioscience's factory in Andong, North Gyeongsang. [JOINT PRESS CORPS]
The government set a budget of 68.7 billion won ($60 million) to support the cost of clinical trials and minimize the number of obstacles on the way to vaccine development, saying that additional funding will be available when necessary. In 2020, the budget was 48 billion won.
It also vowed to purchase vaccines from local companies in advance when they obtain the interim results of phase 2 clinical trials and conduct successful phase 3 clinical trials, considering the immunogenicity, safety, possibility of success, production capacity and ease of injection.
"The government will fully support companies that plan to have phase 3 clinical trials in the second half of the year,” said Vice Health Minister Kang Do-tae. “If Korea succeeds in making vaccines of its own, it will contribute to stabilizing vaccine supply in the country, as well as to overcome the global crisis.”
Kang also said the government would prepare for the post-Covid-19 era by “providing intensive support to secure mRNA vaccine technology.”
Despite the enthusiasm from the government, the jury is still out on whether it is a real and viable goal. It is, after all, a question of money.
The 68.7 billion won of budget to support the cost of clinical trials is far too small, experts say, arguing that more aggressive investment and support from the government is urgently needed.
“That’s a ridiculously small amount of money," said Kang Dae-hee, professor at Seoul National University’s department of preventive medicine. "It cost hundreds of billions of won to conduct phase 3 clinical trials alone. The government set 27 trillion won for R&D investment, but of that, only two trillion won has been allocated to the entire bio industry.”
“Tremendous amounts of money should be put into the bio industry in a very short time [in order to develop vaccines]. Look, the United States injected tens of billions of dollars and it actually succeeded in developing vaccines of its own.”
The United States has spent a total of $10 billion so far investing in the probable vaccine-developing companies. This not only includes U.S. companies Pfizer, Moderna and Novavex, but also Britain-based AstraZeneca and France’s Sanofi.
Kang also added that the shortage of manpower is another problem that needs to be tackled.
“The coronavirus variants will harshly attack the world again soon,” said Kang. “All manpower from everywhere like hospitals and big pharma should be gathered together to develop the biotechnology.”
Lee from KoreaBIO agrees with Kang.
“The pandemic is a really good opportunity for Korea to catch up with developed countries as a latecomer in this area," said Lee. "We need to keep bringing in mRNA technology and trying it out, and the government should fund such efforts and help with networking for global clinical trials.”
"Since there will be such pandemics in the future, Korea must have vaccine technology to prepare for it."
Other Asian countries such as Japan and China are also investing money to become a hub of vaccine production in Asia.
China succeeded in developing vaccines of its own from two companies, Sinovac and Sinopharm. In mid-May, China’s Shanghai Fosun Pharmaceutical Group announced that it will establish a joint venture with the German pharmaceutical company BioNTech SE for the local production and commercialization of mRNA vaccines.
In May, Japan's Nikkei reported that Moderna CEO Stephane Bancel is considering Japan as a possible country for a CMO contract.
BY SARAH CHEA [chea.sarah@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)