[ANALYSIS] Korean Humira biosimilars set for huge 2023
Published: 19 Feb. 2023, 07:00
Updated: 21 Feb. 2023, 10:23
![A Celltrion researcher conducts a test of its biosimilar product at the company's lab in Songdo, Incheon. [CELLTRION]](https://koreajoongangdaily.joins.com/data/photo/2023/02/21/ffdd4ff0-2024-4b89-94aa-cc462d2ba222.jpg)
A Celltrion researcher conducts a test of its biosimilar product at the company's lab in Songdo, Incheon. [CELLTRION]
With Humira, the world’s top-selling drug for over a decade, losing its monopoly, Korean biosimilar makers are gearing up to tap into the approximately $20-billion market.
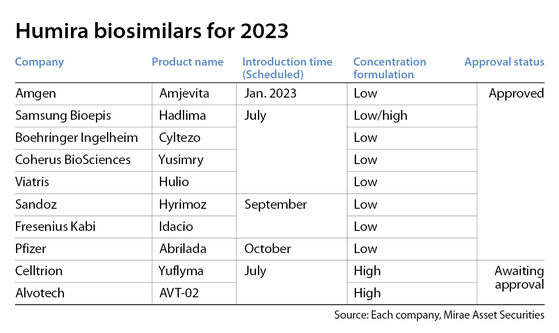
At least 10 biosimilar copies of the autoimmune disease treatment are slated to be introduced this year, including two from Samsung Bioepis and Celltrion.
Big global names also threw their hats into the ring, including U.S.-based Amgen, Pfizer and Germany's Boehringer Ingelheim.
Samsung Bioepis’ Hadlima and Celltrion’s Yuflyma are two promising candidates for the global market with their competitive edges in concentration formulation and interchangeability, two key factors that most other global companies have yet to achieve.
![AbbVie's Humira [NEWS1]](https://koreajoongangdaily.joins.com/data/photo/2023/02/21/c9c73886-9290-46fa-be33-037020213768.jpg)
AbbVie's Humira [NEWS1]
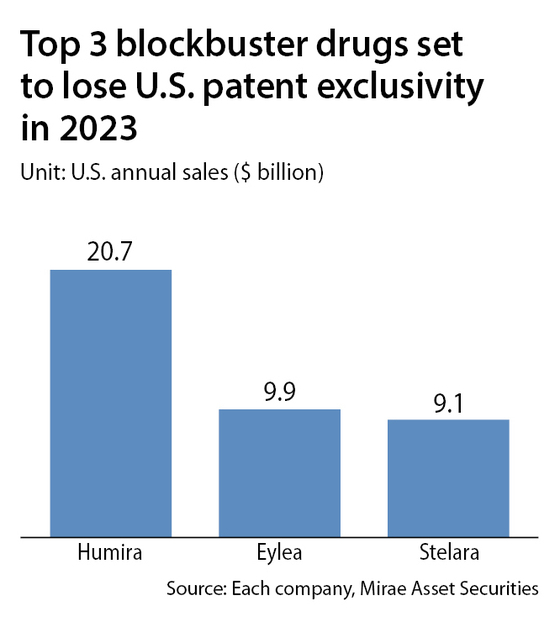
It generated global revenue of $31.6 billion in 2021. Some 66 percent of that revenue, or $20.7 billion, came from the U.S. market, a sum that's equivalent to the entire Korean biopharmaceutical industry.
It had been the world’s best-selling for a decade until 2020, when it lost its throne to the Pfizer Covid-19 vaccine.
AbbVie, Humira's Illinois-based developer, held an exclusive grip on the market for two decades, earning nearly $200 billion from sales of the drug during that period.
“Humira is a historically big event in the biosimilar market,” said Kwon Hae-soon, a researcher at Eugene Investment & Securities.
“If a company succeeds in grabbing just 10 percent of the market, that means they can pocket 1 trillion won ($775 million) every year just with that single drug,” Kwon added. “How large share companies grab in the Humira biosimilar market will eventually determine their global status.”
AbbVie said it forecast a 37 percent drop in sales of Humira this year due to the release of cheaper biosimilars.
Biosimilars, according to the U.S. Food and Drug Administration (FDA), are biological products that are approved based on proof that they are highly similar to other FDA-approved products. The drugs have no clinically meaningful differences in terms of safety or efficacy from the reference product, but they cost less.
High-concentration formulation
Product differentiation by concentration threatens the multiple adalimumab biosimilars approved but not yet launched.
Higher concentrations are generally favored by the market for their convenience as they allow patients to take lower doses and may cause less discomfort. High-concentration formulations account for some 86 percent of the U.S. adalimumab market.
California-based Amgen introduced Amjevita, the first Humira biosimilar in the U.S. market, on Jan. 31 as a low-concentration formulation.
![Samsung Bioepis's Imraldi, a Humira biosimilar marketed in Europe by Biogen . It will be sold under the name of Hadlima in the U.S. market in partnership with Organon. [SAMSUNG BIOEPIS]](https://koreajoongangdaily.joins.com/data/photo/2023/02/21/e1d90e93-80a6-46c4-bd38-bc4d6e778a8e.jpg)
Samsung Bioepis's Imraldi, a Humira biosimilar marketed in Europe by Biogen . It will be sold under the name of Hadlima in the U.S. market in partnership with Organon. [SAMSUNG BIOEPIS]
“Amjevita can be the sole player in the market for about five months until other products are released,” said Kim Tae-hee, a researcher at KB Securities.
“However, Amjevita faces a big barrier as it's a low-concentration formulation, which accounts for only 14 percent of the market."
Samsung Bioepis’ Hadlima is currently the world’s only adalimumab biosimilar approved by FDA as a high-concentration formulation.
Samsung Bioepis also has a low-concentration version, the only company to do both.
When Samsung Bioepis releases both versions in the U.S. market on July 1, the company will become the second to introduce a Humira biosimilar, with local sales handled by New Jersey-based Organon.
Celltrion’s Yuflyma is currently waiting for FDA approval. It is expected to be released in the U.S. market in July.
Yuflyma is already on sale in various European countries. It was the world’s first high-concentration Humira biosimilar approved by the European Medicines Agency.
No other high-concentration candidates from global companies are approved. Luxembourg-based Alvotech has finished Phase 3 clinical trials for its high-concentration candidate and is currently awaiting approval.
Interchangeability
Interchangeability is also a key factor that will contribute to the success of biosimilars.
An “interchangeable label” from the FDA allows pharmacists to choose the biosimilar over Humira when filling prescriptions, rather than requiring the prescribing doctor to specify it.
Amgen’s biosimilar isn’t approved as interchangeable — at least not yet.
Samsung Bioepis and Celltrion are currently undergoing clinical trials for their products to be recognized as interchangeable with Humira.
Samsung Bioepis was initially scheduled to finish the trials in September but advanced it to May. Celltrion is expected to complete the trials in August.
![Celltrion's Yuflyma [CELLTRION]](https://koreajoongangdaily.joins.com/data/photo/2023/02/21/3c229717-b63d-468b-a161-f22e4ba6befe.jpg)
Celltrion's Yuflyma [CELLTRION]
Only one biosimilar — Cyltezo from Boehringer Ingelheim — was approved by the FDA as interchangeable. But, above all, the product is a low-concentration formulation.
What makes companies prioritize interchangeability is that it may help candidates get listed on the formularies of pharmacy benefit managers (PBMs).
PBMs, as third-party companies that function as intermediaries between insurance providers and pharmaceutical manufacturers, create formularies (lists of prescription medications covered by health insurance plans), negotiate rebates with manufacturers, process claims and occasionally manage mail-order specialty pharmacies.
PBMs control more than 70 percent of U.S. pharmacy claims volume, so including a biosimilar product in their formularies can determine its success.
“Major PBMs like CVS Caremark and OptumRx operate many pharmacy chains so they could be generous to drugs that are approved for interchangeable,” said researcher Park Jea-gyeong from Hana Securities.
“It is highly likely that the destiny of Humira biosimilars will be determined by PBMs’ formulary lists.”
Major PBMs including Optum Rx and Express Scripts said that they will include Humira biosimilars in their formularies at parity with Humira.
Optum Rx said it will place the first three biosimilars to reach market on its list, including Amgen’s Amjevita.
“Celltrion’s candidate is definitely distinguished from its rivals in terms of the formulation, so it has the potentiality to be listed on formularies,” said Seo Keun-hee, an analyst at Samsung Securities.
“If Yuflyma can get selected as a preferred drug by PBMs, it will be able to grab some 20 to 30 percent of the U.S. Humira biosimilar market.”
![Samsung Bioepis researchers conduct test at the company's lab in Songdo, Incheon. [SAMSUNG BIOEPIS]](https://koreajoongangdaily.joins.com/data/photo/2023/02/21/8256f3fc-1f7d-45a3-983a-9c02b75302d0.jpg)
Samsung Bioepis researchers conduct test at the company's lab in Songdo, Incheon. [SAMSUNG BIOEPIS]
Other blockbuster lineups
2023 is a big year for biosimilars, with the patents of other blockbuster drugs scheduled to expire.
The U.S. patents of Eylea, an eye treatment made by Regeneron Pharmaceutical, and Stelara, a psoriasis treatment by Janssen Pharmaceuticals, are expected to expire in the second half.
Samsung Bioepis finished Phase 3 clinical trials of SB15, its biosimilar referencing Eylea. It also completed trials of SB17, a biosimilar version of Stelara.
Celltrion is in the last round of trials for CT-P42, its biosimilar referencing Eylea. The Korean company already produced meaningful results proving the efficacy and safety of its biosimilar of Stelara.
Dong-A ST recently announced the Phase 3 clinical trials results of DMB-3115, its candidate referencing Stelara, proving its parity with the original drug.
The size of the total market of biosimilars referencing drugs whose patents are set to expire this year is estimated to be about $33.2 billion. That is the largest figure since 2013, when the biosimilar market started to boom.
BY SARAH CHEA [chea.sarah@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)