Cloning technology breathes life into Jurassic Park dreams
Love or loathe it, cloning technology has evolved in leaps and bounds since Dolly the sheep emitted its first “Baa” in 1997. Who’s that surprised when we read about cloned dogs and cows these days?
But scientists are now speaking about more complex and, to be honest, more squeamish possibilities that raise all sorts of questions, like how would I feel with a pig’s kidneys in my body, and what’s that woolly mammoth doing running around City Hall.
For starters, scientists are getting very excited about xenotransplantation, which means taking live tissues and cells from one species, say a horse, and putting them in a human for medical reasons. Lab boffins are also getting excited about using organs from pigs that are gnotobiotic, i.e. bred in a largely germ-free environment, to save endangered animals and reboot extinct ones.
Xenotransplantation has understandably raised many ethical questions, but one of the benefits is saving lives. A black market for human organs is already thriving, so there’s no doubt about the issue of need.
The pig is the animal of choice for many biomedical researchers interested in cloning for organs since their physiology and anatomy are closest to humans. The goal is to create mini pigs that weigh between 80 to 120 kilograms (176 to 265 pounds) with human-sized organs.
The plan sounds worthy, but there’s a hitch: The human body has a well developed immune system that shuts down anything it thinks shouldn’t be there, like organs from farm animals.
One way to get around our bodies’ paramilitary defense system is to build human chromosomes and genes into the gnotobiotic pigs’ hearts and organs. In theory, this process could suppress rejection.
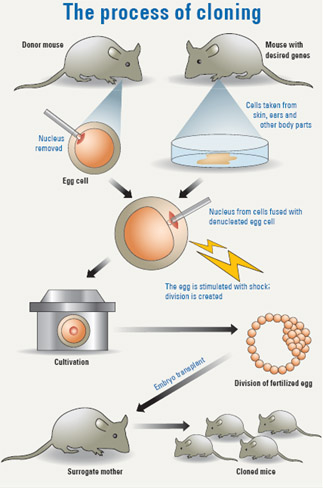
However, Seol Jae-gu of Mgen, a bio research company that has been working on cloning gnotobiotic pigs for the purpose of organ transplants, claims he and his colleagues have solved this problem.
“A cloned gnotobiotic pig without the alpha-gal lining has already been produced in Korea,” Seol claims, adding that more research is needed.
American and European researchers are making progress in cloning gnotobiotic pigs. In 2006, a team at the transplantation biology research center at the Massachusetts General Hospital, U.S., transplanted the heart from a mini pig into a monkey, which survived for six months. And a monkey lived for 83 days after it received a kidney from a pig at the same center.
“In the academic and research field, despite the immune rejection responses, if the organ recipient can survive for about six months, the procedure is considered a success,” said Lee Hoon-taek of Konkuk University’s Animal Biotechnology Frontier.
Seol of Mgen is more cautious, though.
“A successful xenotransplant will take place some day but at this point, we are still trying to control the immune rejection response. As of now, I see the possibility of using xenotransplants to provide short-term solutions in which those in need of organs can use pig organs as a temporary measure until a human organ or proper treatment is identified,” he said.
Another kind of interesting biomedical research involves cloning gnotobiotic pigs for people with diabetes. The pancreas, the organ which produces insulin, contains far less alpha-gal than other organs. Simply put, the pancreas from gnotobiotic pigs has far less chance of initiating the human body’s immune system. In 2005, Mgen cloned gnotobiotic pigs with pancreatic cells that fought off 60 to 70 percent of attacks launched by the body’s defenses. The company is currently trying to make pancreatic cells in pigs completely compatible with humans.
“Xenotransplants are going to become a reality in the near future. Korea has one of the top cloning technologies needed to make xenotransplants possible. It is important at this point to focus more on eliminating the immune rejection response through further research,” said Lee of Konkuk University.
Other cloning projects that have the biomedical community buzzing include dogs that detect cancer. Seoul National University and RNL Bio, a biomedical research company, cloned Labrador retrievers earlier this year that can sniff out traces of cancer on people’s breath.

From top to bottom: A gnotobiotic pig cloned at Konkuk University last year; cloned Labrador retriever puppies that can sniff out cancer; an adult Labrador retriever with similar detective skills. Provided by Konkuk University and RNL Bio
Since 2005, Japanese and Russian scientists have been working on a project to bring to life a woolly mammoth, a creature that died out about 10,000 years ago.
“We have already secured the technology to bring the woolly mammoth back, and we merely need to secure cells with its DNA,” said Akira Iritani of Kinki University.
Scientists are starting to discover mammoth bones and fur in the defrosting Siberian tundra as ice melts due to rising global temperatures. The well preserved carcass of a six-month-old female was discovered in northwest Siberia recently. Iritani estimates that a cloned baby mammoth will be born within 50 years at the latest.
The most difficult step in bringing extinct animals back from the dead involves securing a well-preserved nucleus from somatic cells, which form the body of any living creature. But recently a Japanese scientist cloned a mouse using brain cells from mice that had been frozen at minus 20 degrees Celsius (minus 4 degrees Fahrenheit) for 16 years. So the technology exists.
The next step is to secure an appropriate donor and surrogate animal. The researchers on the mammoth project believe Asian elephants would do the job because of biological similarities between the species.
Seoul National University’s research team led by Shin Nam-shik was the first to clone a wolf, an endangered species, last year.
“Maybe the same logic doesn’t apply to dinosaurs or mammoths that have been extinct for many years, but cloning endangered animals such as polar bears and Siberian tigers among others is possible,” said Shin.
While cloning can help extend human lives and save endangered species, Ku In-hye, a professor at the The Catholic University of Korea’s department of medical and life sciences, thinks there’s a need to look at the root of the problem before we seek technologically advanced solutions.
“Before we start cloning endangered or extinct species, we need to think about why these animals are dying, or have died out. It’s usually because of destruction of the environment and natural habitats caused by humans. Once cloned, can we realistically expect the animals to survive in an unnatural habitat?” Ku asked.
But there’s one cloning question that has everyone hooked: Do we have the technology to clone humans and is it going to happen?
But that’s a whole new story, perhaps the most controversial ethical dilemma facing the human race.
At the moment, isn’t the thought of woolly mammoths quite enough to contemplate?
By Ko Sung-pyo, Kim Jung-soo JoongAng Sunday [jason@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)