[Venture Abroad] DNA tests still face legal hurdles
Published: 21 Aug. 2016, 18:34
The country’s Bioethics and Safety Act restricted analyzing individual gene information to hospitals and clinics, saying the tests violated people’s dignity and could result in unexpected consequences. Authorities argued that “gene test results vary depending on which institution rolled out the test, and ultimately, an inaccurate test result can lead to unnecessary confusion.”
Because of such restrictions, gene analysis in Korea has only been used by medical institutions in legal cases such as confirming paternity.
In June, the Ministry of Health and Welfare lifted regulations on 46 types of genes that can provide results on 12 health indicators, allowing companies as varied as pharmaceutical and bio-related start-ups to engage in DNA testing.
The 12 health indicators include cholesterol rate, blood pressure, possibility of hair loss, skin elasticity, response to caffeine and more.
Despite the eased regulation, industry experts say there are still too many restrictions preventing Korea’s gene-testing industry from catching up to global standards.
“The 12 tests that the government has allowed will have a minor effect on the local bio industry because it doesn’t include critical indicators that people are really curious to know through their genes such as the possibility of cancer in the future or the origins of their personality traits,” said one industry insider who asked not to be named.
“What makes a real difference in the market and will invigorate it are the DNA tests that are still in the hands of clinics and hospitals.”
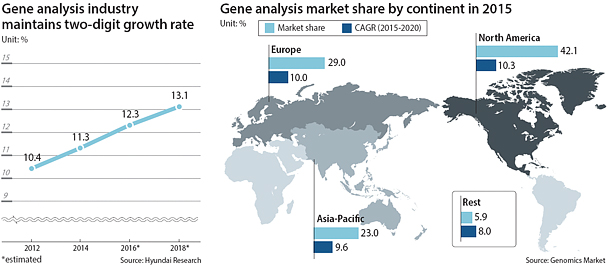
The global gene-testing market is expanding at a rapid pace, maintaining annual two-digit growth since the early part of the decade. Its size is now estimated at around $12.3 billion, and by 2018, the industry is expected to grow to $13.1 billion, making it comparable to the global semiconductor market, which was valued at $10.57 billion last year.
23andMe, a Silicon Valley start-up that analyzes DNA through saliva, is valued at $1.1 billion, with subscribed users topping one million last year, according to The New York Times. Shortly after the company was founded in 2006, though, it was faced with regulation hurdles from the U.S. Food and Drug Administration (FDA).
Co-founder Anne Wojcicki told The Times that the rate of new subscribed users dropped by half after the service was temporarily suspended by the FDA in 2013. The regulator raised concerns that the DNA test results could be medically inaccurate.
Similarly, medical experts in Korea believe commercializing individual DNA tests requires more time.
“Non-medical personnel may misuse the DNA information to exaggerate results, and if an error has occurred in the evaluation process, there is no legal way to restrict them,” said Kim Joo-hyun, a spokesperson for the Korean Medical Association.
BY JIN EUN-SOO [jin.eunsoo@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)