Celltrion share price shoots up
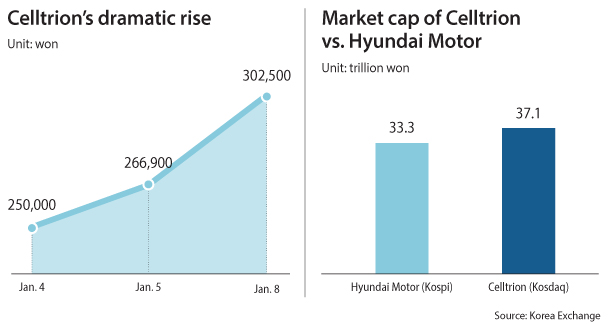
The share price of Celltrion, which is listed on the tech-heavy secondary index Kosdaq, rose by 13.34 percent, or 35,600 won ($33.46), on Monday compared to the previous session to close at 302,500 won, after it gained about 17,000 won from Thursday to Friday.
As a result of the drastic price hike, the market cap of the pharmaceutical giant reached 37.1 trillion won Monday, exceeding the market cap of Korea’s biggest automotive company Hyundai, which currently ranks at third on the primary Kospi with a market cap of 33.2 trillion won. Hyundai closed Monday at 151,000 won per share, up 1.34 percent from the session on Friday.
According to market analysts, the recent Celltrion rally is a result of the improving investor sentiment that the company’s biosimilar products will quickly gain a market share in Europe as well as in the United States, the world’s biggest pharmaceutical market.
Kang Yang-koo, an analyst from HMC Investment Securities, said that the market share of Truxima in Europe stood at seven percent as of the third quarter last year, outpacing Remsima in terms of speed of market penetration.
Truxima is a biosimilar used in the treatment of a number of diseases, including rheumatoid arthritis and non-Hodgkin’s lymphoma. Remsima is a biosimilar that treats autoimmune diseases such as rheumatoid arthritis, inflammatory bowel diseases and Crohn’s disease.
Kang also projected that the market share of Remsima in the United States, where the company received permission for its sale from the U.S. Food and Drug Administration (FDA) in April last year, will surpass 15 percent this year.
“The rapid rise of the share price is also reflective of the expectation that Herzuma will be released in Europe without any problem and quickly gain a share in the market just like the other drugs,” explained Kang.
Herzuma is a biosimilar version of trastuzumab, a breast cancer and gastric cancer treatment antibody drug known by its brand name Herceptin, patented by Swiss drug firm Roche. It is likely to gain approval by the European Medicines Agency (EMA) and go on sale in Europe during the first quarter of this year.
“After Europe, we expect that Celltrion will gain approval for Herzuma and Truxima by the U.S. FDA in August or September,” the analyst added.
Still, other analysts explained that the market is taking uncertain future events into consideration prematurely and that it is possible that there will be a dramatic price fluctuation in the near future.
“The positive market sentiment we saw early in the year is mainly centered on the drugs that are yet to go on sale,” said an analyst who requested anonymity.
BY CHOI HYUNG-JO [choi.hyungjo@joongang.co.kr]










with the Korea JoongAng Daily
To write comments, please log in to one of the accounts.
Standards Board Policy (0/250자)